 Click
to see a larger image.
Click
to see a larger image.SUMMARY
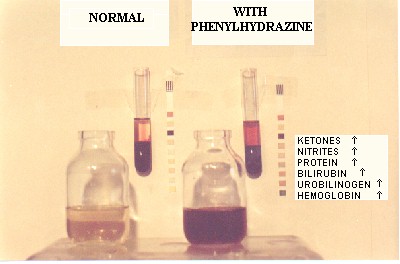
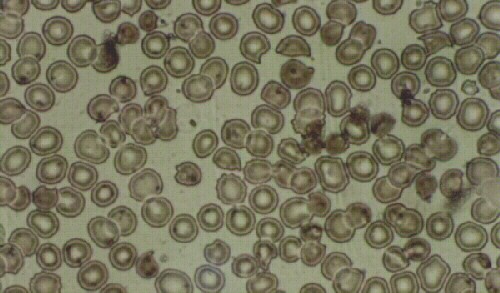
Patients with Chronic Mountain Sickness or Excessive Erythrocytosis (EE) as it is currently called, are permanent residents of high altitude 3600 m, and have red blood cell counts (RBC) above 6.5 x 106 and appear cyanotic. This causes aesthetic and psychological problems in their life. Occasionally, observers may believe they are alcoholics. When a hemogram reports increased RBCs, they search for a miraculous cure. According to several previously evolved concepts of EE, treatments have included: leeches, marrow radiotherapy by administration of radioactive substances like phosphorous, and more recently, phlebotomies, tea infusions, garlic tablets and most dangerously, administration of a previously banned cytotoxic agent, phenylhydrazine. One concept of providing treatment for EE patients is to permanently destroy their RBCs. However, the effect of phenylhydrazine usage is toxic for the bone marrow, the liver and other tissues, changing the color of the skin from cyanotic to yellow, with jaundice. The conjunctivae of the eyes become icteric and the urine becomes dark brown. Once started on treatment, patients are tested periodically and their RBC count indeed diminishes, and they may feel satisfied. However this toxic medication may have a fatal outcome. By reducing the number of RBCs, the arterial oxygen content (CaO2) of the blood is diminished. We have found the majority of patients with EE to have abnormal chest x-rays. Ergometric testing of these patients may produce severe oxygen debt. In one of the patients, on the 4th stage of the Bruce Protocol, intense pain in both legs gradually became intolerable and he required post-exercise oxygen. Following interruption of the phenylhydrazine, CaO2 returned to normal in approximately 60 days, with a rise of RBCs slightly above previous levels. The patient experienced no symptoms of illness. This case and multiple other cases have lead us to believe that EE is a compensating mechanism for lung disease at high altitude and that the RBC mass should not be decreased.
TRATAMIENTO INADECUADO DE LA ERITROCITOSIS EXCESIVARESUMEN
Los pacientes con el mal de montaña crónico o eritrocitosis excesiva (EE) son residentes de la altura (3600 m) con 6.5 x 106 glóbulos rojos (GR) o mas y presentan cianosis. Esto ocasiona problemas estéticos y psicológicos en su vida ya que las demás personas creen que son alcohólicos. Cuando un hemograma reporta aumento de los GR, ellos buscan una cura milagrosa. De acuerdo a los conceptos evolutivos de la EE, los tratamientos incluyeron: sanguijuelas, radioterapia de la médula ósea mediante administración de substancias radioactivas como el fósforo, y mas recientemente, flebotomías, infusiones de te, tabletas de ajo y la mas peligrosa la administración de la fenilhidrazina, agente citotóxico prohibido. Encontramos que la mayoría de los pacientes con EE tienen placas radiográficas de tórax anormales. El concepto de los tratamientos es el de disminuir los GR. Sin embargo, la fenilhidrazina es tóxica para la médula ósea, el hígado y otros tejidos, cambiando el color de la piel de cianótica a icterica. Las conjuntivas se tornan ictéricas y la orina café oscura. Una vez iniciado el tratamiento, la sangre de los pacientes es analizada periódicamente y el recuento de GR disminuye, con lo que quedan satisfechos. Sin embargo, esta medicamento tóxico puede producir la muerte. Al reducir los GR, el contenido arterial de oxígeno (CaO2) en la sangre disminuye. Las pruebas ergométricas en estos pacientes durante el tratamiento producen gran débito de oxígeno. En el paciente descrito, en el 4to nivel del protocolo de Bruce, el dolor intenso de ambas pantorrillas se hizo intolerable y requirió oxígeno post ejercicio. Al interrumpirse la fenilhidrazina, el CaO2 retorna a niveles normales en aproximadamente 60 días, con una elevación de los GR por encima de los valores iniciales, y mejoría de la capacidad de ejercicio. Este y muchos otros casos nos llevan a creer que la EE es un mecanismo de compensación de la enfermedad pulmonar en la altura y que la cantidad de GR no debe ser disminuida.
INTRODUCTIONSince Monge (2,3,4) described chronic mountain sickness (CMS), the treatment of these patients has been controversial. These high altitude inhabitants suffer from an increased number of red blood cells and are cyanotic. They usually are diagnosed with Excessive Erythrocytosis because they consult a physician due to their cyanosis or headaches, or simply because a routine blood test reveals an increased hematocrit. Frequently they are told that their high hematocrit is very dangerous and so their understood objective is to find a doctor who can lower the number of red blood cells. Unfortunately, they may fall easily into the hands of unscrupulous physicians. Hence they receive multiple panaceas or see physicians who propose phlebotomies. However some physicians have given these patients, phenylhydrazine, a hemolytic agent long abandoned because of its toxicity (1). In the past, we have seen several cases treated with this medication.
We present a case report in order to illustrate the toxic effects of this drug in a patient with EE. He was initially seen by us, before being given the treatment and later while he took the drug.
METHODS
Patient FG was first seen in consultation because of cyanosis, headaches and fatigue. He was a non-smoker, drank alcoholic beverages occasionally and had minor digestion problems. His father died of lung cancer and his mother had a cardiac pacemaker. Five brothers were healthy. A complete physical examination was performed on this 44 year old. Weight was 74.3 Kg, height 163 cm, blood pressure 120/90 and his pulse was 90 per minute. The electrocardiogram revealed elevation of S-T segments in V-2 and V-3. On laboratory exam, the hematocrit was 67 %, hemoglobin 22.3 gm%, 7.5 million red blood cells per mm3, and he had 8700 white blood cells per mm3 and a 70 % neutrophil differential count. The urine analysis was normal.
Pulmonary function studies found a diminished forced vital capacity (FVC), 79% of predicted, FEV.1 77% predicted and FEF 25-75 59% of predicted. A flow-volume curve was compatible with chronic bronchial obstruction. The Nitrogen Washout test revealed a diminished Total Lung Capacity (87% predicted), and residual volume (72% of predicted), with no signs of uneven ventilation. Radial artery blood gases at rest found a PaCO2 of 39 mmHg, PaO2 39 mmHg and pH of 7.38, a compensated respiratory acidosis. His chest x-rays, showed enlarged hila, nodular images distributed in both lungs, a 2 cm in diameter calcified nodule, and an enlarged heart.
A hyperoxic test breathing 100 % oxygen showed significant pulmonary shunt reaching a PaO2 of only 104 mmHg (normal above 200 mmHg). He was diagnosed as having chronic obstructive pulmonary disease, cor pulmonale, myocardial ischaemia and pulmonary shunting with compensatory excessive erythrocytosis. He was given antibiotics, mucolitics, bronchodilators, and a weight loss diet.
 Click
to see a larger image.
Click
to see a larger image.
DISCUSSION
This is another case of the previously reported iatrogeny (5). This
patient was diagnosed as having EE secondary to long term cardio-pulmonary
disease at high altitude (3600m). When he was originally treated by us,
we focused on improving the pulmonary function and his hematocrit increased,
but so did his PaO2 (52 mmHg). While receiving Phenylhydrazine his oxygen
content was reduced dramatically and due to the large shunt, his PaO2 was
41 mmHg, although he looked less cyanotic (actually yellow). The exercise
test found him capable of achieving the 5th level (normal, for a sedentary
healthy person at 3600 m) but at great oxygen debt. This was a patient
with a large intra-pulmonary shunt whose compensating EE was significantly
reduced. Therefore the oxygen content of his blood was reduced along with
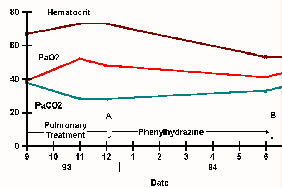
his work capacity. In another patient, the arterial oxygen content gradually
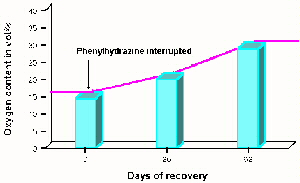
increased in 62 days, upon interruption of Phenylhydrazine and it was observed
that there was a significant improvement on uphill walking the steep streets
of La Paz, something he had been unable to do while on the drug (fig. 5).
 Click
to see a larger image
Click
to see a larger imageOur previous experience on patients with EE found no one with the intense post-exercise cyanosis and oxygen debt that this patient experienced. Although unable to reach level 5, their recovery is usually uneventful. We strongly recommend that patients with EE and pulmonary disease, not be treated with this drug, nor should they have their RBC mass reduced. To our knowledge there is no safe drug treatment for reduction of EE. Treatments should be directed at the etiology of the pulmonary lesions that cause EE at high altitude, not at the secondary increased red cell mass.
BIBLIOGRAPHY